Hygiene
Superabsorbents
Superabsorbents are a key component of hygiene products like baby diapers or adult incontinence products, ensuring that urine is locked up safely, keeping the skin and clothes dry and clean.
Today’s disposable diapers are high-tech products, which have become even more comfortable, thinner and absorbent over the years. A major contributing factor to this development has been innovative superabsorbents such as HySorb® and SAVIVA® from BASF.

How superabsorbents work and how they improve our lives
Superabsorbents are humble in appearance: In the shape of inconspicuous white particles they are mostly hidden in the core of diapers, adult incontinence and hygiene products and therefore generally out of sight. But their effect is impressive. Superabsorbents absorb and retain large quantities of liquid even under pressure. Superabsorbents are instrumental in ensuring that diapers work effectively and that the skin remains dry and healthy.
A superabsorbent experiment -
how superabsorbents work
One gram of superabsorbent can absorb up to 500 grams of pure water. The liquid is not only absorbed but also locked up inside the particles even under external pressure. The result is a soft gel that is still capable to absorb further liquid.

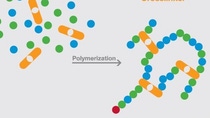
The chemistry behind it
Superabsorber is made by combining (polymerizing) single molecules of sodium acrylate to form long molecular chains. The polymerization is activated by an initiator, and a crosslinker connects the single molecules together to form a polymer network. The crosslinker ensures that the granules remain insoluble when exposed to liquid, maintaining their absorbent properties and structure. Upon contact with aqueous liquid the sodium ions become dissociated generating an osmotic pressure which drives more liquid into the superabsorbent binding it tightly within.

Development of diapers since 1980
Superabsorbents enable modern diapers to be thin, extremely light and very comfortable to wear. In the mid-1980s, before superabsorbent polymers were introduced into diapers, much more absorbent fluff pulp was needed to absorb a similar amount of urine. Baby diapers were thicker; at just under 100 grams they weighed around three times as much as they do today and could retain significantly less urine. Without superabsorbents, liquid could be squeezed out of the diaper, for example by the baby putting pressure on the absorbent core when sitting down. With a typical SAP amount of around 12 grams, today’s modern baby diapers prevent urine leakage, stay dry for hours and protect the skin from moisture helping to avoid diaper rash.
Download your information on HySorb®

